|
ALL
THINGS KOI AND H2O
Viral Gill Disease
Introduction
Many folks will remember the funzies we had
last year with a potent bug variously called:
· Bacterial Gill Disease (BGD)
· Viral Gill Disease (VGD)
· Fulminate Gill Rot (FGR)
· Necrotic Gill WhatChaMaCallIt (NGWCMCI)
Where this bug hit, it always caused staggering losses. Entire ponds
were wiped clean in a matter of days. Whole collections were lost. Some
very "advanced" ponders who generally would laugh at most of the common
bugs suffered staggering losses. Having played with this bug myself
and having been buried in piles of dead fish for more than 4 months,
I can tell you its a real mover. A few folks deny that it exists at
all, but I'm here to tell you otherwise. Fortunately we're seeing only
a few scattered cases emerge this year, but it's far from certain this
bug is really gone. We might be seeing the tail end of this disease
process... or we might be seeing the build-up of a massive epidemic.
All speculation and political posturing aside, nobody knows for sure.
All we can do is wait, load our guns, and pray we never have to use
them.
In the meantime, a few folks have been tracking this disease and working
on treatment options. In the past this disease was considered completely
untreatable. We think we've made substantial progress in this area,
but understand our work has been "meatball" to put it mildly. Due to
the fast-moving nature of the disease, just getting infected samples
for culture often involves jumping through hoops and all sorts of monkey-motion.
Many times these "infected" fish turn-out to have nothing more than
a bad case of flukes, ick, or other common pathogen. But in other cases
we've seen a complete lack of traditional bugs and still witness massive
morts. These are the cases we've lived for.
Where actual cases of VGD were found to exist, we had a better than
70% recovery rate if the treatment was applied immediately upon onset.
It must be stressed that the purpose of this paper is to bring to light
the problem, relate its symptoms, and disclose certain treatment protocols
we've developed to combat it. This paper is not to be taken as a call
to panic. It is my personal opinion that 99% of all the ponders in the
world will never have need of this information and can view this paper
as a technical curiosity. But for those who are visited by this bug,
knowledge is indeed power. We have demonstrated again and again that
this disease can be stopped if caught early, treated aggressively, and
the procedures herein followed carefully. To those folks who believe
they have this bug: It must be stressed that 95% of the "Viral Gill
Disease" cases we have seen turned-out to be something else after careful
analysis. Anyone who looks at a pond full of sick fish and pronounces
"Yep! They've got VGD!!" is completely and utterly milking without a
bucket. As of this writing, VGD is still rare. It is only when all other
avenues of explanation for epidemic losses have been exhausted that
the thought of VGD should be seriously entertained. If you think you
have VGD, contact any of the individuals at the end of this document.
Finally, a cautionary note on the treatment protocols described herein:
Because of the nature of this disease and due to the lack of any successful
"scientific" effort within the koi community, we have resorted to the
piscine equivalent of "battlefield medicine". The treatments we propose
herein, effective as they are, are most definitely of a last-ditch nature.
These treatments can kill fish. The dosing is absolutely critical. The
difference between killing the bug and killing the fish is often only
a 5% or 10% difference in dosing. These protocols are not for the chemically-disinclined
or feeble-minded.
Do not begin these protocols until all other possibilities have been
systematically exhausted and you are left with no choice. This is clearly
a case where the cure can be as hard on the fish as the actual disease.
I would like to extend my personal gratitude to the author of this document,
Spike Cover, for his continuing efforts to effect a cure as well as
to Doc Conrad and Duncan Griffiths for their support, testing, and refining
of the therapies.
General conditions, Fortunately this disease
has not become wide spread this year (as of June 2000). However sporadic
cases have been reported this Spring in both Britain and the US. Two
variations of the disease have been observed. One seems to activate
at a temperature in the mid-70°s F, the other about 10°F cooler. The
warm water variation seems to run its course in about two weeks, i.e.,
most of fish that are going to die, do so within about two weeks of
the first death. Deaths are slower but steady and ongoing in the cold-water
variation, lasting over a month or so. The disease has almost always
occurred as the pond water is moving into a particular temperature range,
i.e., 65°F to 78°F. Both variations are more prevalent as the temperature
is on an upward trend. Fish exposed to the disease become symptomatic
about 2 weeks after first exposure if the temperature is in the susceptibility
range of 65°F to 78°F. Injectable antibiotics of any type seem to have
no effect on the progress of the disease (which suggests the pathogen
is viral in nature).
The disease frequently occurs within a few weeks of the introduction
of new fish.
Also frequently (but not always), the new introductions are the first
to die.
Symptoms include: ·
The affected fish exhibit a general listlessness (much more so than
usual). · Some fish, the ones that are affected the worst, begin hanging
in the water; usually head down and not swimming much. This is also
a symptom of gill flukes and other parasites that affect the gills.
Microscopic examination should be employed to eliminate parasites as
the cause of the gill function impairment.
· Affected fish collect near a source of water that's well oxygenated.
· Some fish having a blotchy appearance on their skin (usually a reddening)
· Some fish having patches on the body that are rough to the touch like
sandpaper and like the mucous layer is gone in that area.
· Some fish may have patches of what appears to be skin/scales sloughing
off - particularly on the head.
· Some fish may exude a heavy gray mucus that is unevenly distributed
about the body.
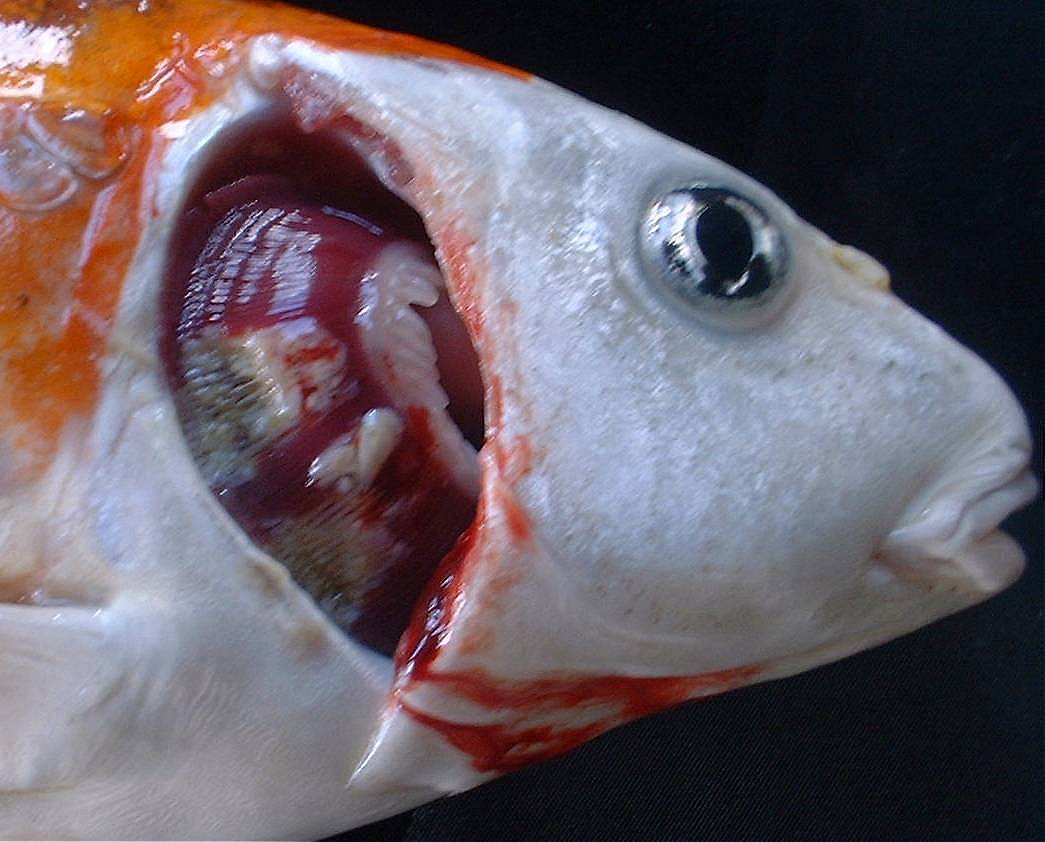
· On inspection, the gills of most fish appear necrotic (dead tissue
usually appearing whitish or tan in streaks or patches). However, a
small percentage die w/o obviously necrotic gill tissue or other external
lesions.
· The eyes become very sunken in the advanced stages of the disease.
· As disease advances, 5% to 10% of the fish die each day - less in
the cold-water version.
· The disease usually hits the larger fish hardest and first. They lay
on the bottom with their pectoral fins spread (other fins clamped) as
if to keep from rolling over.
· Not all fish moralities are alike in the initial stages. A small number
die with no signs of gill damage and no external lesions. However, as
the disease gathers momentum, gill degeneration becomes rampant.
· Pathology reports on diseased fish suggest that the gill damage is
secondary to the viral infection with the virus weakening the subject
to such a degree that it additionally falls pray to opportunistic pathogenic
bacteria in the environment.
· It should also be noted that while small numbers of ecto parasites
may be present upon microscopic mucus examinations, they are typically
deemed to be not significant contributors to the onset of the disease.
In some cases, there may be no ectoparasites present.
Treatments: Chloramine-T ("C-T") (Developed
by Roark and Dr. Erik Johnson in 1999 and revised through additional
testing by Roark 1999-2000.
This is the most completely tested and successful of the treatments.
Roark recovers 70%-80% of affected fish using this treatment if caught
early.) Theory of the chemical treatments (C-T and PP): The virus weakens
the immune system and its ability to combat the opportunistic bacteria
that invade the fish in its weakened condition. The bacterial infections
are likely secondary to the viral infection. The chemical treatments
kill most of the bacterial on the fish and in the water. Additionally,
Roark theorizes that C-T reaction products enter the fish's system thru
the gills (one of the most affected organs) and passes out of the fish
via the kidneys and the liver (two other organs typically affected by
the disease). Further, if the secondary bacterial infection has infected
the liver and/or the kidneys, the C-T may help kill the bacterial invaders
in those organs. Day 1 - Take the fish off their food. This disease
affects the kidneys and liver and they'll need these organs untaxed
by wastes just to survive. Treat the pond (including all the fish in
it) with potassium permanganate ("PP") sufficient to keep the water
pink/purple for 6 hours (1 to 3 ppm depending on the strength of the
fish) then immediately move all fish to a quarantine tank if possible
and treat with about 25 ppm equal; to 9.2 gm per 100 US gal. of C-T
for 4 hours (see the following Note).
Then do an immediate massive water change (80% or more). Note: that
the therapeutic dose is only slightly less than the lethal dose, and
these doses are very greatly influenced by pH and the water hardness.
Note also that the above dose is perfectly acceptable with a pH of 8.0
to 8.5. The dosing table at http://www.koivet.com/chloraminet.htm
Note: J. Spangenberg's article on flukes and C-T. See Table 1 below
which can be used as a rough guide but Roark found it was too conservative
in dosing levels for this disease. He recommends that the level be raised
to where the fish begin to tox-out, then do a 15-20% water change to
dilute the dose a bit. If you did your guessing right, they'll still
be slightly toxed for the duration of the treatment, but they'll make
it. Again, this treatment is not for the meek.
Table 1: Doses of Chloramine-T with respect to water
pH and hardness
|
pH
|
Soft Water Dose (mg/L) |
Hard Water Dose (mg/L)
|
|
6.0
|
2.5
|
7.0
|
|
6.5
|
5.0
|
10.0
|
|
7.0
|
10.0
|
15.0
|
|
7.5
|
18.0
|
18.0
|
|
8.0
|
20.0
|
20.0
|
|
8.5
|
25.0
|
25.0
|
From the above chart, it becomes obvious that treating
at a pH of 8.0 or above is desirable as the hardness of the water becomes
insignificant. Note: adding baking soda to water tends to buffer the
pH to 8.4.
Day 2 thru 5 - Do nearly the same C-T/water-change treatment except
reduce the C-T dosage by 10% to 15% of the rate determined on Day 1.
If the fish show signs of overt toxicity, execute a small water change.
Day 6 thru 10 or 12 - Once the fish are on the mend, don't hesitate
to run 1 ppm PP for 6 to 8 hour a day if you think you need to "clean-up"
the water and keep it sterile.
Do not feed the fish to soon after the treatment as the kidney an liver
will be working overtime clearing up the mess left from the virus/bacteria
and do not need the extra load from feeding. Wean them back to food
slowly and lightly after 10 days or so feeding high quality food (such
as ShoKoi ImpactÔ) sparingly. After treatment, if you lose fish due
to massive gill damage, there is an experimental protocol Roark has
used which doses a 3% H2O2 solution at 0.2 ml per US gallon every 12
to 18 hours combined with a one-time "standard" dose of methylene blue
("M-B"). The H2O2 provides "chemical" oxygen support (1-2 ppm O2 gain)
and also tends to kill some bugs at the same time, while the M-B provides
an alternate means of getting O2 into these fish. M-B is used here as
an oxygen transport mechanism. This is a poorly understood effect but
it works and indeed is documented in some of the (non-koi) literature.
Use it only if you have clear signs of mortality due to impaired gill
function. Do not use M-B in combination with the PP discussed above
for days 6 thru 10 or 12. A water change to eliminate the M-B prior
to treatment will be required.
AlsoNote that excessive use of H2O2 as an oxygen support mechanism is
to be discouraged. H2O2 is a oxidizer which, if overused, will contribute
to gill failure and necrosis. Note too that the use of salt is a good
idea, but it is neither reasonable or necessary to exceed 0.3% salinity
with this disease. This salt level is used to help relieve the osmotic
pressure difference the fish must contend with between its body and
the surrounding water, not for any real bug-killing effect. It is also
useful to run a 5% to 10% (of total salt dose by weight) mixture of
*potassium* chloride. This further unloads the fish and provides potassium
(a secondary electrolyte) that may help with stress. Also note the above
dosing rates are for US gallons. (Imperial gallon = approx 1.2 US gallons)
Recommendation:
If the local tap water temperature is lower than 65°F, it is suggested
that a two-tank system be used so that the water in the holding tank
and filter can be left intact and heated while treatment is under taken
in the second tank. Extra O2 should be placed in the treatment tank
during the treatment. After the 4 hour treatment is complete, the fish
can simply be placed back in the untreated water of matched temperature.
This effects a 100% water change. The treatment tank can then be emptied,
filled and heated to the holding tank temperature in preparation for
the next day's treatment. Warming the water to 70° to 75°F is desirable
because very little tissue regeneration takes place below about 65°F.
Heat Theory of heat treatment:
Many viruses have temperature limits beyond which they are not active
or will deconstruct. Many bacteria are also adversely affected by high
temperature. When the temperature is raised above the upper threshold
of the virus' preferred range, the koi's immune system is better able
to cope with the virus and many of the pathogenic bacteria as well.
Heat treatment would need to be done in the early stages of the disease
since the oxygen saturation level of water is lower at higher temperatures
making it more difficult for compromised gills to absorb adequate oxygen.
It has been reported that placing the koi in water at a temperature
of 30°C (86°F), will aid the fish in beating the disease (this bug does
not like heat). This was reported on the KoiVet web board in December
of 1999. This post claimed that the Israelis had intentionally infected
their koi with the viral disease and then put them in 30°C water where
many recovered w/o any further treatment. Potassium Permanganate There
have been several reports of fish being cured with potassium permanganate.
The reports indicated that the treatments were at 1 gram/100 gallons
for 4 hours for 5 consecutive days, then repeated again after 3 days
(see: http://www.koiusa.com/research/vanjen.htm)
Contributors The following persons were instrumental in developing this
document and treatment methodology:
Name EMail Contribution Area Roark roark7@aol.com Chlor-T, M-Blue /
H2O2 protocol development
Griffiths.D duncan.griffiths@ntlworld.com Chlor-T, M-Blue / H2O2 protocol
validation
Conrad, R. docconrad01@aol.com Chemistry Advisor
Cover, S. scover@pacbell.net Disease history,
Document author Copyright © 2000 by S.Cover, D.Griffiths, R.Conrad and
Roark. All Rights Reserved. Email comments to: roark7@aol.com Page rev
1.10 of 28JUN2000
|

